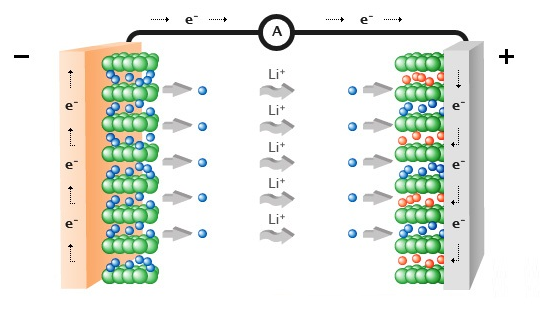
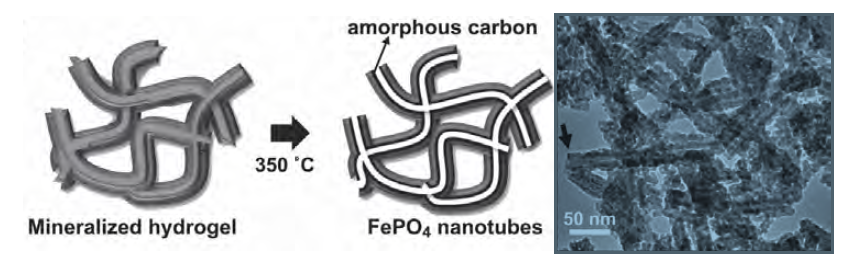
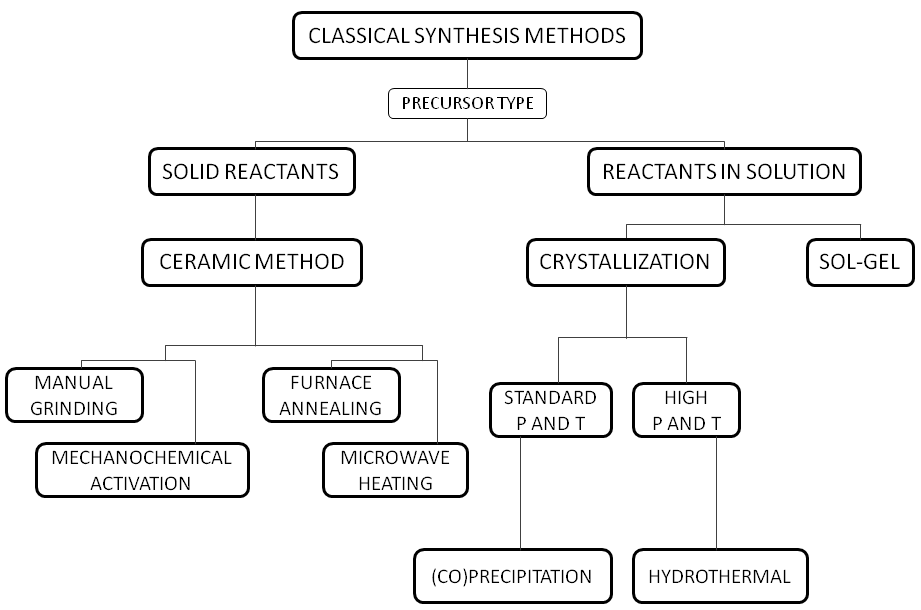
UN38.3 refers to paragraph 38.3 of the "United Nations Manual of Tests and Standards for the Transport of Dangerous Goods" specially formulated by the United Nations for the transportation of dangerous goods. The safe transportation of lithium batteries must have a test report that meets the requirements of UN38.3 and a certificate. The cargo transportation condition appraisal report that co...
Learn More English
English  English
English français
français Deutsch
Deutsch русский
русский italiano
italiano español
español 日本語
日本語 Polski
Polski svenska
svenska 中文
中文





 IPv6 network supported
IPv6 network supported